Services
List of services provided by CF BIC.
Measurements are performed by CF BIC staff after obtaining samples from users. The speed of service depends on chosen technique and current workload of CF staff, however, the results are typically available within a week.
Since 2021, the services are requested via booking system.
Biomolecular characterization and interaction:
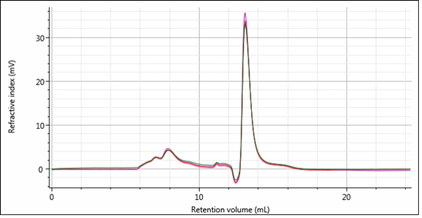
Analytical SEC-MALS
Analytical size-exclusion chromatography coupled to an array of detectors including UV-VIS spectrometer, refractometer, viscometer and static light scattering (RALS/LALS). Suitable for analysis of sample purity, oligomeric state of proteins, study of complexes and other experiments.
For more details, contact the CF responsible person, Monika Kubíčková.
e-mail: monika.kubickova@ceitec.cz
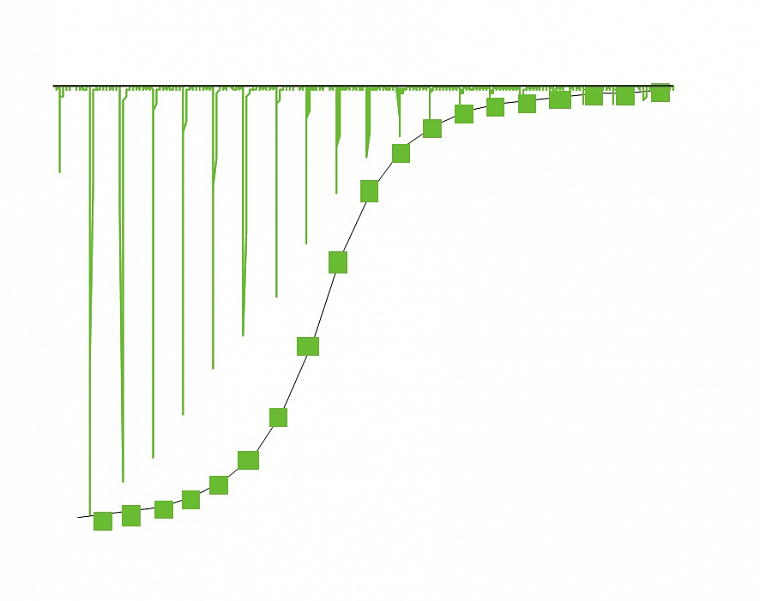
Sedimentation velocity method can be used for determination of sample dispersity, oligomeric state of proteins, the shape of the particle and it is also suitable for analysis of aggregates and a study of biomolecular interactions.
Sedimentation equilibrium method can be used for accurate determination of molecular weights of various biomolecules and the study of their interactions.
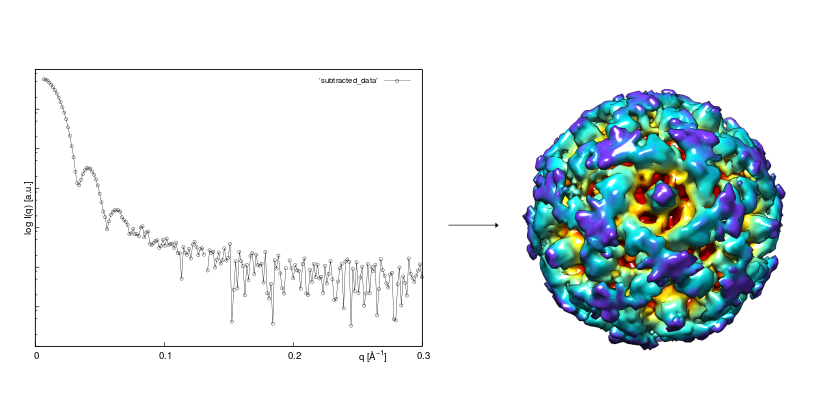
Biological Small-angle X-ray scattering
SAXS is used for the determination of the integral structural parameters, shape reconstruction, determination of the oligomeric state in solution, unraveling of the quaternary structure of protein complexes, and modeling of molecular flexibility. CF BIC offers SAXS data acquisition and SAXS data analysis services.
Contact the CF responsible person, Tomáš Klumpler, for details.
e-mail: tomas.klumpler@ceitec.cz
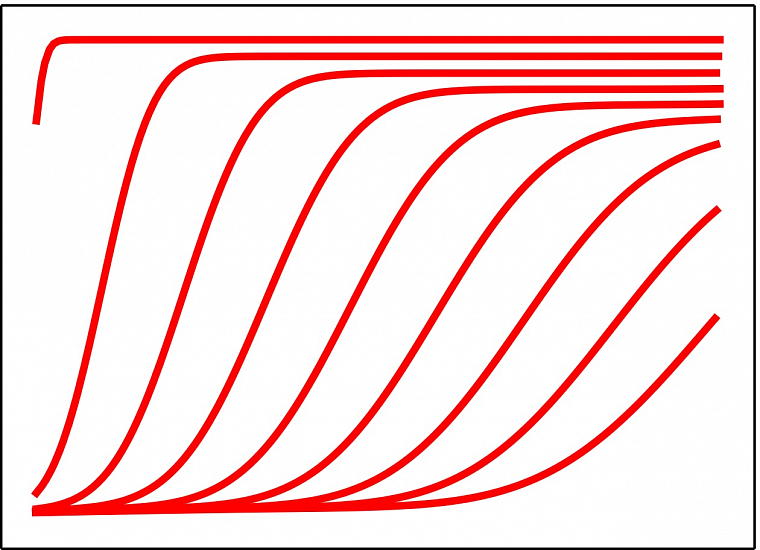
Calorimetric measurement of receptor-ligand interaction (Standard titration method, Single injection method) Data evaluation - by automated software or manually by expert - complete thermodynamic profile determination (association constant, enthalpy, entropy, stoichiometry value) using curve fitting models: One set of binding site, Two sets of binding sites, Sequential Binding model. Determination of kinetic parameters Km and kcat.
Differential scanning calorimetry
PEAQ-DSC is differential scanning microcalorimeter with <200 µl volume of sample capillaries. DSC technique measures the heat changes that occur in the (bio)molecule sample solution during a controlled increase or decrease in temperature, on the basis of a temperature difference between the sample and the reference material. Automated version of PEAQ-DSC enables to run up to 282 samples in a row unattended.
AUTO-PEAQ
DSC
Sample quality assessment
Sample quality service enables to characterize the protein sample prior to more advanced experiments. It includes measurement of intact mass (MS, in collaboration with CF Proteomics), aggregation content (DLS), and temperature stability (nanoDSF).
MS
nanoDSF
DLS
Protein production services:
We offer protein production services, including cloning and preparation of expression systems in both E. coli and insect SF9 cells, followed by solubility screening and large-scale expression. Our downstream capabilities include protein purification using IMAC, SEC and/or StrepTRAP affinity methods, ensuring high-quality yields. A minimal quality check package is also available to verify protein integrity and suitability for further applications.
Offered services:
- Cloning
- E. coli - expression system preparation
- E. coli - solubility screening
- E. coli - large scale expression
- Insect cells - expression system preparation
- Insect cells - large scale expression
- Protein purification - IMAC affinity
- Protein purification - SEC
- Protein purification - StrepTRAP affinity
- Minimal quality check (package) - DelsaMax and TwoMP
For more information on Protein Production services please contact radka.dopitova@ceitec.cz
Crystallography of biomolecules:
Set-up of crystallization screens (drop volume 200-300 nl) using commercial kits (including kits for membrane proteins, protein complexes and nucleic acids) or multidimensional gradients for optimization (from up to 5 different solutions). Sitting (standard) or hanging drop crystallization method, crystallization under oil. Storage of the screening plates for 3 months at a constant temperature 4°C or 20°C. Regular automatic inspection within the period of one month. UV imaging to distinguish a protein from a salt. Optional storage of the screening plates for an extended period of time (>6 months) at a constant temperature 4°C or 20°C, with a possibility of on-demand extra inspections
Optimization of protein crystal production in large scale (manual setting), Membrane protein crystallization techniques, Crystallization by dialysis or using capilaries. The service price is calculated individually. Contact the responsible person for details.
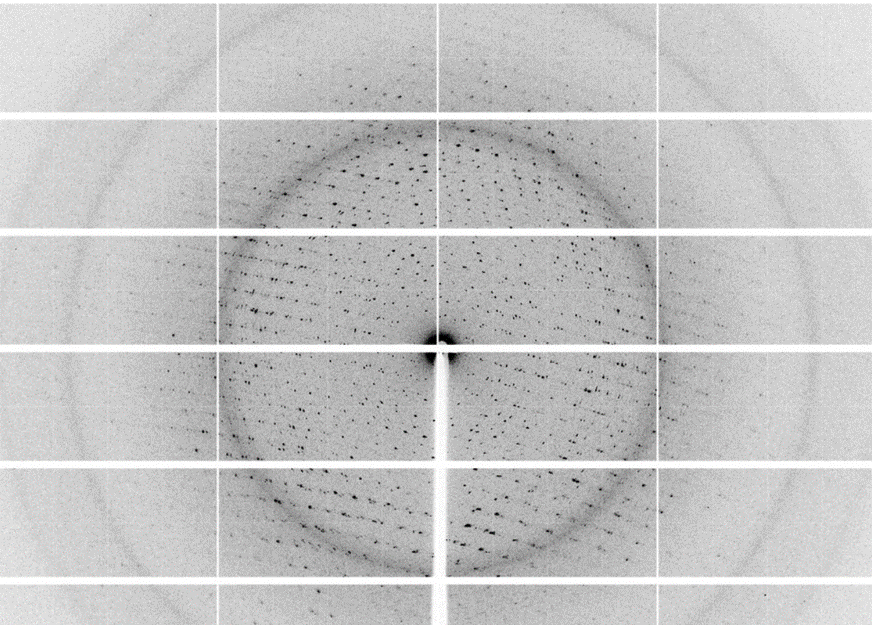
Using a home X-ray source, the diffraction of crystals can be measured. Various modes from testing of diffraction quality up to complete data collection and structure interpretation are available. Contact the responsible person for details.
Initial training for instruments in user mode:
Initial training for instruments, which are operated by users him/herself. This includes Octet RED96e (BLI), Jasco J-815 (CD), SpectroLight 600 and DelsaMax Core (DLS), Biacore T200 (SPR), multichannel SPR imaging system, Auto-PEAQ DSC and VP-DSC, Prometheus (DSF), VP-ITC, Monolith NT.115 Nano and Monolith NT.115 Pico (MST), Biacore T200 (SPR), multichannel SPR imaging system, microdeposition system for biosensor arrays, gradient incubator for optimizing protein crystallization temperature (TG-40) and Biospectrometer kinetic (UV-VIS KINETIC)
Training is performed by CF BIC staff - the responsible person for each instrument. Without this training, users cannot operate the instruments on their own.

Other:
Service includes consulting with specialist, expert data evaluation, advanced experimental design or personal user support during measurement.
Please, contact responsible person for details.
Material (consumables)
While basic consumables are provided by Core facility for free, specialized material can be ordered. This includes SPR and BLI sensors, MST capillaries and fluorescent dyes or material for protein crystallization. For more details, contact CF staff.
CAPILLARIES
BUFFER SCREEN
LABELING KIT